Research
Our interdisciplinary approach to research runs the gamut from discovery to application. We embrace collaboration and team science—so we can swarm quickly around real problems as soon as they arise.
Research Strengths
Latest Research News
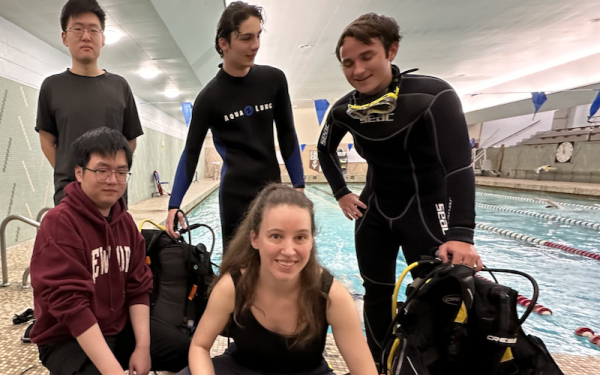 Monday, April 15, 2024
Monday, April 15, 2024 Kathryn Daltorio's Crab Lab tests underwater robot in Veale Center swimming pool
 Friday, April 12, 2024
Friday, April 12, 2024 Research team led by Case Western Reserve awarded $2.75M from Department of Defense to advance clot-stabilizing nanotechnology
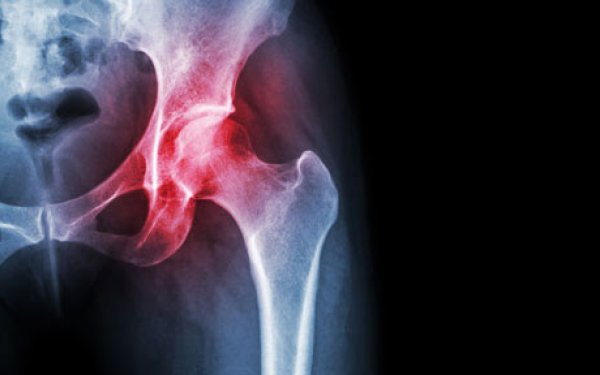 Wednesday, March 27, 2024
Wednesday, March 27, 2024 Case Western Reserve University awarded federal contract to develop and commercialize ‘live’ replacement joints






















![exterior of Sears think[box]](https://engineering.case.edu/sites/default/files/styles/thumbnail/public/subsite_images/Sears-thinkbox-exterior.jpg?itok=VhGtVmwW)

